Solutions for Researchers
Our NK-92® cell line provides a versatile bioanalytical testing solution that helps researchers develop functional killing assays that are more consistent and reliable than donor blood and reporter gene assays.
We license NEUKOPANEL® CELL LINES – A standardized panel of CD16 expressing NK-92® cell lines that provide for the first time the means to establish a consistent, robust, and reliable assay for assessment of ADCC activity.
Brink Biologics, Inc., a ImmunityBio, Inc. affiliate company, serves as the exclusively-licensed distributor of ImmunityBio’s proprietary NK-92® natural killer cells, and offers a variety of GLP and research-grade, NK-92®-based cell lines as part of its Neukopanel® portfolio of products for laboratory testing applications. Neukopanel® includes Conkwest® cells for discovery, translational, and developmental research as well as commercial lot release testing of clinical products. Brink products are utilized widely by large pharmaceutical and biotechnology companies to support products such as anti-viral and dendritic cell vaccines, therapeutic monoclonal antibodies and biosimilars, bi- and tri-specific fusion proteins, cytokines, natural killer cell checkpoint inhibitors, and other biologic products. These Neukopanel® cell lines bring the unique enabling technology behind ImmunityBio’s platform to the broader drug discovery and development community.
ImmunityBio’s NK-92 cells are highly valued by researchers because they preserve the NK function while providing ease of access, propagation, maintenance, and utility for both bench and in vivo applications. Their use as an effective natural killer cell surrogate has been demonstrated in more than 450 published papers covering a wide range of research and testing investigations. NK-92 cells have emerged as the de facto cell line of choice for relevant academic and commercial innate research. In addition, NK-92 cell-based assays have become the trusted and increasingly popular choice of large pharma and biotech companies for their growing antibody development programs and commercial product release testing.
Five advantages of Neukopanel® cell lines and solutions for researchers:
Neukopanel® cell lines provide the means to establish consistent, robust, and reliable assays for assessment of true ADCC function with these five advantages.
Cost and Time Savings
Cost and time savings over common practice using donor-sourced (PBMC) NK cell isolation, expansion, and maintenance in short-term cultures.
Eliminates phenotypic variability
Eliminates phenotypic variability of donor-sourced NK cells, as Neukopanel® cell lines offer clonal consistency, purity, and reliability as an assay reagent.
Ease of Use
Ease-of-use over donor-sourced NK cells, and capable of long-term continuous cultures.
Assay Robustness
Ability to generate a readout spectrum using our homozygous high- and low-affinity CD16 expressing lines alongside our negative control line for assay robustness.
True Cytotoxicity
True cytotoxicity rather than cell-binding readouts.
A Neukopanel® of NK-92 Cell Lines Suitable for a Range of Laboratory Testing Applications
 aNK research grade cells
aNK research grade cells
Our parental NK-92 cell line for product development and research applications lacks CD16 expression and can serve as an effective control for our haNK CD16-expressing cells that are useful for ADCC assays and other applications. These cells also express activating cell surface innate receptors and can be used to explore fundamental NK cell function and cytokine release for drug discovery and product development applications.

haNK research grade cells
Our haNK cells are available with either high or low affinity CD16-expression and are available with or without GFP. haNK cells provide an NK-92 cell line solution for true target cell killing ADCC assays – all without the downsides, costs, inconsistencies, and inefficiencies of reporter gene assays and PBMC donor blood based approaches.
Neukopanel-driven Antibody-Mediated Killing Assays
haNK cells are natural killer cells engineered to incorporate a high-binding affinity receptor (CD16) that binds to an administered antibody and demonstrates its cancer cell-killing effects.
These antibody-targeted haNK cells can directly bind to IgG1-type antibodies, such as avelumab, trastuzumab, cetuximab, and preclinical stage antibodies under investigation to benchmark the cancer-killing efficacy of existing therapeutic antibodies and enable the development of new ones. haNK cells demonstrate this effect through Antibody-Dependent Cellular Cytotoxicity (ADCC).
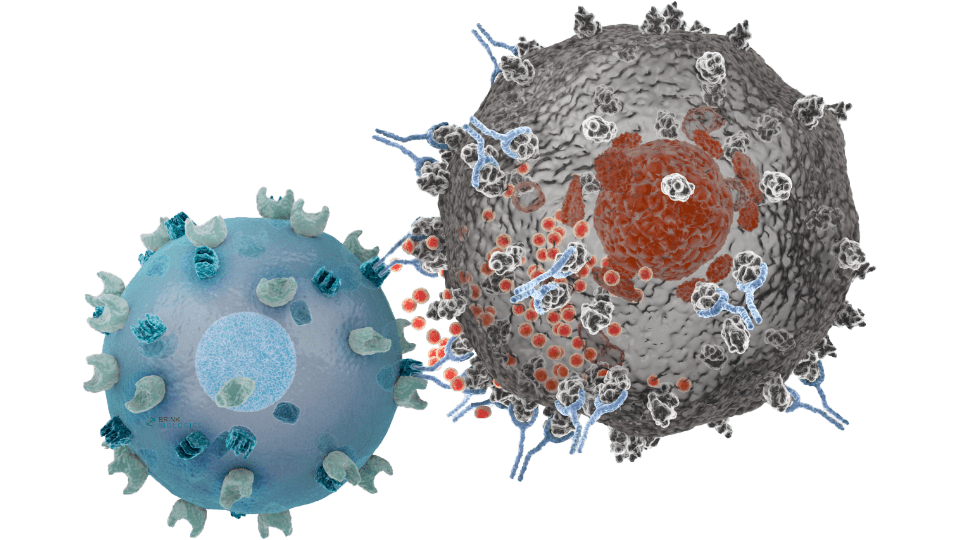
Antibody products are abundantly utilized to treat cancer and are estimated to generate over $100 billion in reported annual sales. A growing number of studies suggest that clinically meaningful responses to these antibody therapies correlate directly with the overall health of a patient’s natural killer cell population, and whether they express the high-affinity variant of the CD16 receptor. Current literature estimates that only 10 to 15 percent of the eligible patient population carry high-affinity CD16 receptors.1. This suggests that our haNK cells with high-affinity CD16 and their complementary low-affinity CD16 variant – both part of Neukopanel – provide a range of CD16 variant options that allow for the generation of physiologically relevant data to help researchers understand the potential of their therapeutic candidates.


